The biohack.me forums were originally run on Vanilla and ran from January 2011 to July 2024. They are preserved here as a read-only archive. If you had an account on the forums and are in the archive and wish to have either your posts anonymized or removed entirely, email us and let us know.
While we are no longer running Vanilla, Patreon badges are still being awarded, and shoutout forum posts are being created, because this is done directly in the database via an automated task.
Engineering yeast to produce spider silk
https://youtube.com/watch?v=Fx8TcGrCOSI
Video sums it up well. This is what I'll be working on for the next while.
Comments
-
Excited for updates on this! Fingers crossed the yeast doesn't give you too many problems.
-
Oh I'm sure they will haha. Already got a list of potential roadblocks and working on solutions in case we hit them. Ought to be a fun project though
-
They aren't the easiest to work with, especially for such a big protein, but I'm sure you'll get it working! When are you planning to transfect?
Also, side note, but you should really consider becoming a professor.
-
Couple weeks. Last of the supplies should arrive this week, then we'll start on the cloning and plsamid assembly. Should be ready to attempt transfection shortly thereafter. First things first is making sure the species of spider I receive is the same as the gene sequenece I have. If not gotta identify and modify/reorder 1 pair of primers. Should be fine, but we'll see. After that gonna find out if the species of yeast I have matches the integration sequence. Again, should be fine, but I'm planning for the worst case. Other than that the rest should hopefully be smooth sailing. Gonna be doing the first PCR to isolate the g418 resitance probably tuesday and then gibson it into the plasmid later in the week. Just finishing up my new heat block to make that process easier. Once that's done we'll try our first transformation to see if the empty plasmid works. If it does it's spider time
Haha I'm flattered. Not sure which uni would hire me though XD I like teaching via my channel. All the fun of teaching, without needing to mark exams XD And it lets me regularly publish null results.
-
Update number 1: https://youtube.com/watch?v=8mnj5evjMX4
Ran our first PCR reaction and Gel and something seemed weird. No pcr product was generated, but stranger still the lane that was supposed to have the donor DNA in it was empty in both runs. Makes me thing that the vial of donor DNA that I got from the odin is just water with no actual DNA in it. Would explain why my attempt to follow the GFP yeast transformation protocol didn't work. Gotta do more tests to confirm but something seems weird. Gonna extract some DNA from the glowing yeast I have and try again. Hopefully that fixes it. If it doesn't then there's either an issue with our derpy little pcr machine, primers or reagents. Gonna test everything and see where the issue is.
-
That definitely sounds like a primer issue to me. Could it be that, as you mentioned, the sequence you received didn't match up with what it was supposed to be and primers didn't actually cut anything?
-
Maybe, but look at those gels. There's supposed to be ~20ng and 100ng in both of the GFP lanes. Those should be the brightest thing on that gel since each band of ladder is about 27.5ng. I'm gonna test the donor DNA plasmid solution with a nanodrop to make sure there's actually DNA in there. Assuming it is we can rule that out and focus on the primers and reagents. We're going to use the fancy PCR machine to do touchdown PCR with a gradient (decreasing annealing temp every cycle). We're using massive primers so we can add gibson sites to the ends of the fragment so the annealing temp is like 53C but the full primer temp is 72c. So hopefully with the gradient we'll hit a sweet spot where it'll anneal properly and amplify. If the DNA is actually missing then I'll do the extraction first and try the PCR again.
Oh also we were using old reagents for the test run. Used an old ass vial of Deep vent polymerase. I've got some nice hifi Q5 coming in the mail this week which ought to be worlds better. Also gonna order DNTPs and stuff so I don't need to use that shitty wax nonsense. I hate that stuff so much. Getting the wax off was such a pain in the ass. It solidifies the second you go to pipette it off.
-
I watched both of the videos you posted. Maybe this is getting a bit ahead of things, but have you considered how codon optimization (or lack thereof) of the spider silk sequence might affect expression in yeast?
-
Ya but since it's impossible to synthesize the gene, there's nothing I can really do about it unless I build the gene from individual silk monomers which is super tedious
-
Another update:
https://www.youtube.com/watch?v=LOyBYEks550Tl:dr, The Odin sent me water instead of DNA. Going to have to extract some from the preengineered yeast.
-
- Lol that's hilarious. Did they properly compensate for the monumental fuck up?
- Never heard of wax before, is it similar to DNTPs?
- A little unrelated, but at the end of your video you mentioned trying out new colors. I found out during my work that mCherry's beta barrels actually result in a more stable protein than most other fluorophores. Not sure what you're doing next, but felt worth mentioning
-
@misslitty
1. No cause it was a free kit meant to be reviewed... on video. So...
2. the wax contains the DNTPs. It's called easy start pcr in a tube. It's a mix of all the usual stuff with some wax so that when the wax melts it forms a protective layer and the aqueous stuff separates out and drops to the bottom. I'm really not a fan though. Getting that wax out was a giant pain in the ass and multiple times we thought we'd fucked the tiny amount of liquid at the bottom before realizing the wax had just solidified... again. You basically have like 10 seconds to work with it and the second the pipette touches the wax it freezes again. God damn nightmare to work with.
3. I've heard similair things from friends working with other beta barrels. the red ones are just way more stable. I'm trying to build up a collection of chromoproteins so I can have the whole spectrum to play with. Also planning some mutation stuff to make more colors. Gonna do mutation PCR with manganese in the reaction to make it lossy. You don't happen to have any of that mCherry plasmid anymore do you? Cause I'd love a sample. Once I get the silk working we're gonna throw beta barrels on the end to make colorful version. I love the idea of a blue sweater that's just like that without adding dye. Beyond that gonna sub out the gfp for rfp in the yeast plasmid and make a red christmas brew, though I dunno if it'll be ready in time. -
@chironex
Oh I suppose that may have backfired a bit. Is the wax at least more affordable?
And that is so cool! Have you played around with manganese before? Also, I'm sorry but I don't have any of the plasmid, worked on project that in a different country. I'm so stoked for this spider silk experiment, though, and can't wait to see what you do with it. -
@misslitty
To be honest I bought it cause I misread the listing. I thought there was Taq in there too. It was cheap though. But I'm gonna get proper dntps and buffer. Found a cheap supplier so gonna give them a shot.No worries, figured that'd be the case but couldn't hurt to ask.
Hehe I've got so much planned for that silk... Shooting for end of November for first silk. Fingers crossed the lab God's play nice.
-
@chironex
Haha live and learn, I suppose. I suspect having separate DNTPs and all will make your life a lot easier.Sending good research and spidey vibes- keep updating us on this please!
-
@misslitty we found a paper that claims to be able to extract PCR quality DNA using nothing but a piece of filter paper. We're trying it with both our yeast and a spider and trying the PCR's on the result with proper reagents tomorrow or the day after. If this works we'll be like 1/2 way done and it'll be the worlds easiest DNA extraction. Will keep ya'll updated.
-
Did the extractions last night. Did 3 spider and 3 yeast. The yeast were from some plated yeast on selection media, and then some from our halloween brew. All should contain the donor GFP plasmid so hopefully 1 of the tubes comes back positive and we can use it for PCR. We're running 1 set of PCRs tonight and I'll be running another tomorrow. As to the spiders we'd been talking about the issue of the silk gland as we figured as soon as we pop the spider the remaining silk would make a mess of our reactions. Can confirm. So we did 3 spider extracts from 2 spiders. the first was just a whole spider, shoved in a tube and mushed. The second was split in half and an extraction was preformed on just the thorax, and just the abdomen. The silk as predicted ended up gooping everywhere but it kinda balled up. I used the standard genaid mini plasmid extraction kit and just used the reagents that came in the kit with no modifications. If this all fails I'll try again but I'll freeze/thaw the spiders and throw in a bit of pepsin to help break the cells open. In the end I had 6 tubes of DNA solution. In theory anyway. Gonna run a gel tomorrow and see if we see anything and then test everything with a nanodrop to better quantify things.
Tl:DR. Squished some spider and some yeast to acquire their essence. Now to isolate the genes we want and build our plasmid.
-
Oh, we also seperatly tried the paper extraction method. That's why we're doing 2 sets of PCR. 1 with the paper method and 1 with the standard column method. May end up retrying both if they dont work. Will keep updating as things progress.
-
Ok so exciting updates and interesting updates.
First the less good which is that of all the DNA extractions I did, they all yielded only very very small amounts of DNA. This is likely because the kit I used was meant for ecoli, so the digestive enzyme in the lysis buffer won't work properly on eukaryotic cells. Gonna retry with some proper protease as well as a freeze/thaw cycle or two and a longer incubator time to make sure things are good and lysed. So stay tuned on that front.On the PCR front we got some results back. First thing is that we successfully isolated the G418 gene. The funny part was that it was from the tube of DNA from the Odin not one of the paper based extracts. SO apparently there was some DNA in there, just an order of magnitude or two less than what was on the label. So probably 1ng/ul or less. Still god aweful for yeast transformations, but sufficient for PCR. Seems the issue with our first PCR run was the crappy reagents. We got new reagents now and things seem to be going better.
Moving on, last night we ran more PCRs on the extract I prepared so hopefully some of them work even with the low levels of DNA. Gonna give it the old college try and if the tiny whiff of DNA in the odin kit was sufficient, hopefully the extracts will be as well since they actually do read postive for DNA. Gonna do a second set on the fancy machine tomorrow and we'll see how it all goes. If it all fails we'll try again one the improved extracts and if that still fails we'll redesign our primers to remove the gibson ends to make it easier to isolate the gene from the genomic DNA.
And finally huge news about our yeast which is i found a cheap source of the correct species of yeast so now we shouldn't have any issues with integration whcih is what i was worried about. So we'll have that shortly as well. So once the plasmid is built we'll stand a much much better chance of producing silk.
Tl:dr 1 gene down, 1 to go. Next week we clone the isolated g418 gene into the plasmid to make our new standard empty yeast plasmid which will be released for people to use. Should have the silk gene shortly as well and the finished plasmid.
-
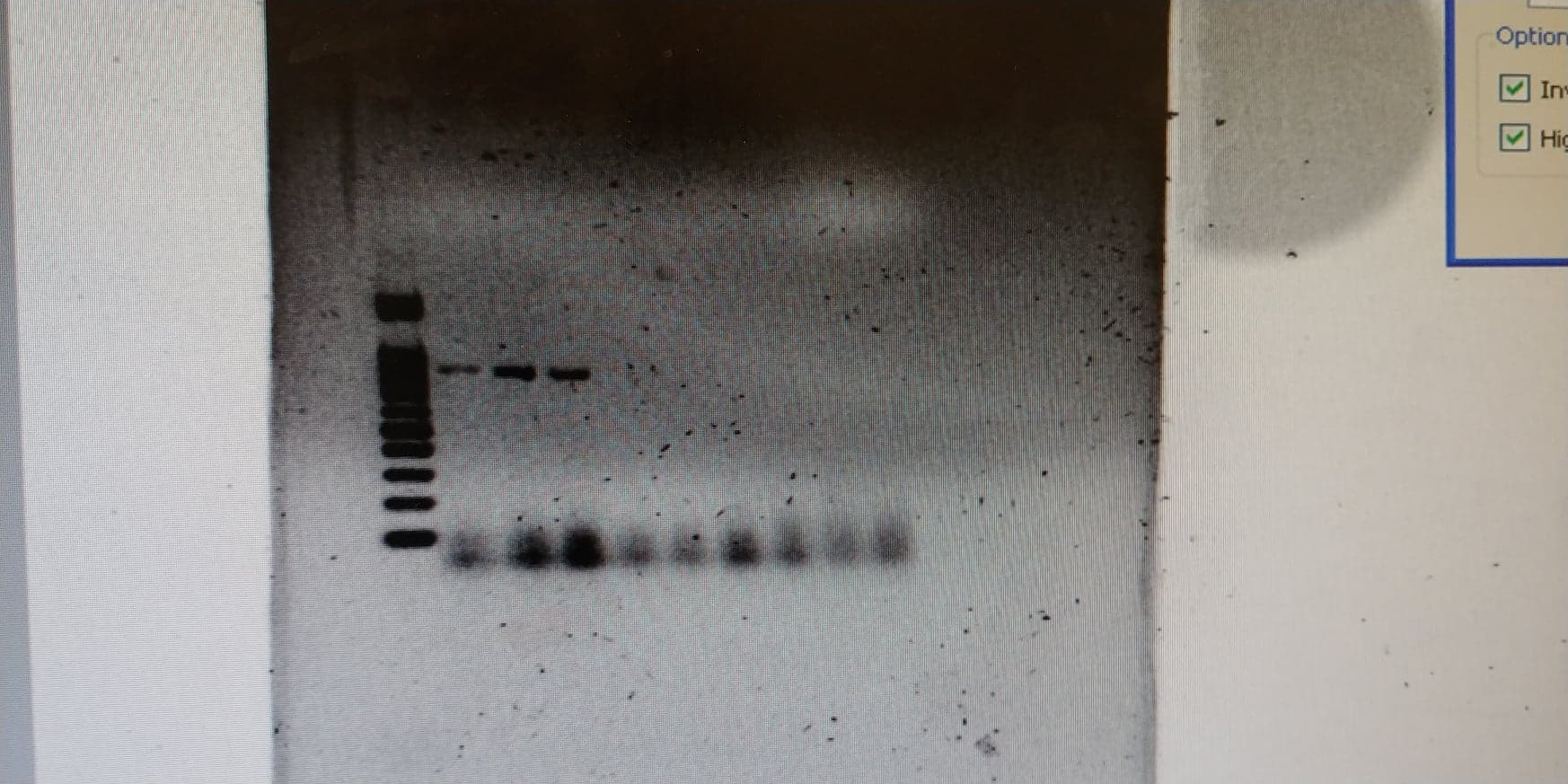
-
What's in what lane? And does that mean the paper-based extraction didn't work?
In any case, very exciting!!
-
The three that worked are all from the same thing just at three different annealing temps. Though the cycle was run with a gradient decreasing in temp each cycle by 1.2 degrees. So in order it's 72, 68, and 65c.
But ya paper based didn't work but since neither did column extraction we suspect it's an issue with our lysis procedure not the extraction method itself.
But yes exciting!!It's funny, the more times people comment saying that I'm not a professional and so stand no chance of making this work when people better than me have tried, the better I feel every time a step works. Was reading the comments under the recent hacakday article on the project and people claim it's impossible to get the strength of normal silk and then reference those stupid goats. I'm excited to mop the floor with them XD as if having a degree is what makes genes express properly ahaha
-
Did someone order a big load of fuck all?
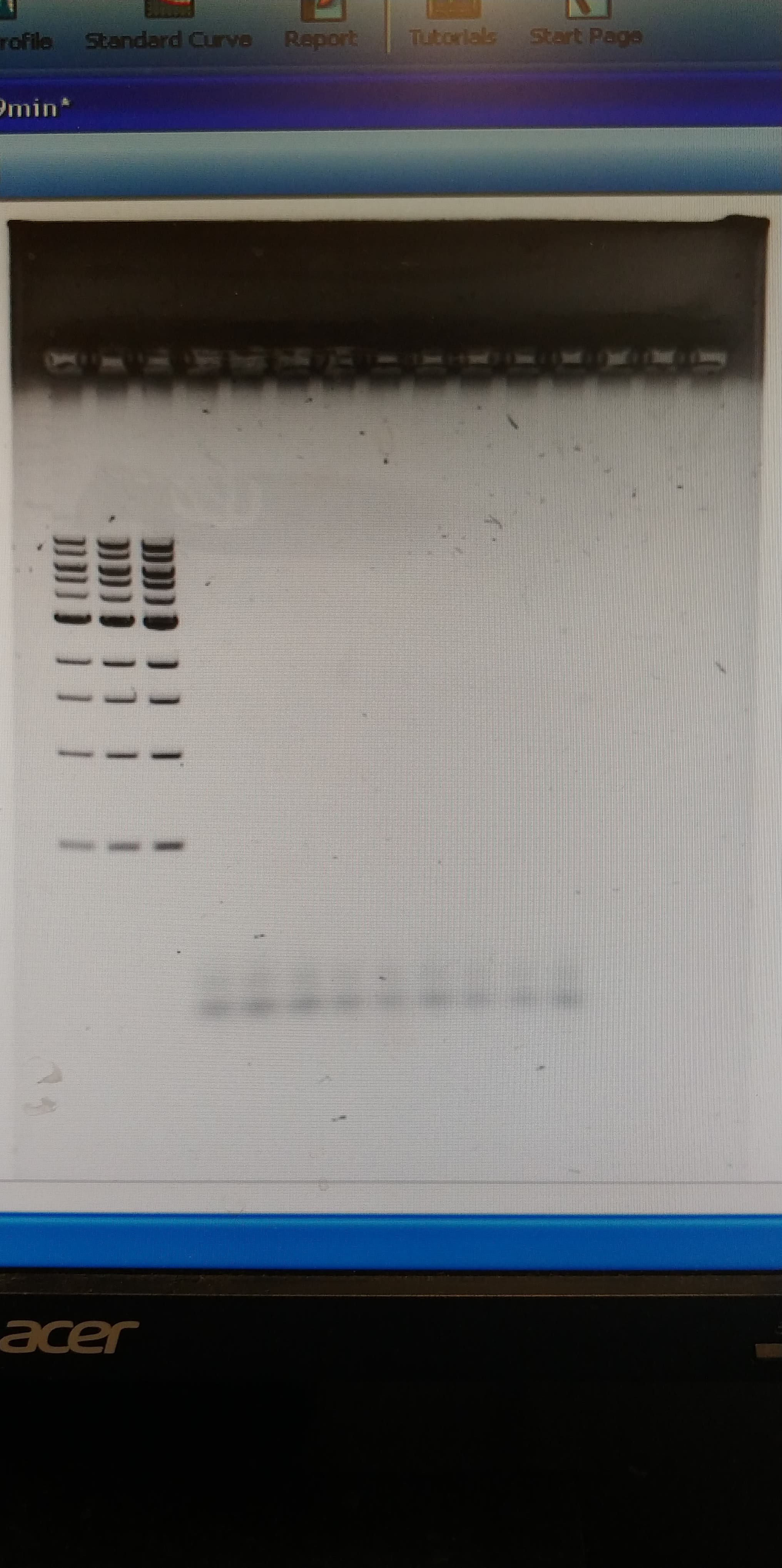
We think the issue was our terrible DNA extraction. Gonna redo that with the new extraction procedure, then try again once we've confirmed we have lots of genomic DNA to work with. If that fails we'll redesign the primers. And if that fails... well I dunno what we'll do but we'll figure something out. We weren't really expecting this to work, it was more of a shot in the dark to see if we'd get lucky. Oh well. Back to sciencing!
-
Secondary update, it's becoming very obvious why our DNA extraction failed. I mean we knew we were using the wrong kit to start with, but proper insect extraction calls for incubation in lysis buffer overnight at 56c rather than the 4 minutes at rt we did for attempt 1. This is very good news! Means come Tueday we should have lots of high quality DNA (and hopefully I never need to handle more of these god forsaken spiders)
-
Latest update and part of the series! https://www.youtube.com/watch?v=7kJ2o7P8D00
-
Some updates. We tried a weird PCR reaction directly on a piece of spider tissue but it didn't work. HOWEVER another run we did on a reaction that should have worked, didn't. Which means my machine might just be crap. So we're gonna run a reaction that we know works and see if it's an issue with my machine. We'll also try the direct spider pcr again on a better machine.
Next week we'll be doing our first restriction digests and gibson assembly to build our empty plasmid.
Also on the latest DNA extraction we did actually get some spider DNA but everything was suuuuper sheared. So I'm gonna run it again with an even better protocol that's more gentle. Everything was so sheared that we didn't have any pieces bigger than 2k which would explain why our pcr didn't work. So hopefully further improvements to DNA extraction technique will sort the problem out. We're also ordering shorter primers to help the PCR go easier. And we may try running 1 run with a GC enhancer coctail to help deal with the difficulty of the sequence.
-
Update time.
Tried more extractions. None worked properly. Showed nice peaks on a nanodrop, but garbage on a gel.
Ordered more spiders, they just arrived, as well as a proper DNA extraction kit. So gonna give that a go hopefully this week.
As to the plasmid we planned on putting everything into, we struck out hard. The sample of it we thought was good turned out to be fucked, and it's impossible to get more since NEB discontinued it and only sells it as a very expensive kit.
The new focus has become getting the silk gene itself. Once we manage that we'll find a new plasmid to put it into. Not ideal, but shit happens sometimes. So gonna run the DNA extraction, and then PCR the result with shorter primers and hope for the best.
I'm officially frustrated.
end of update.
-
Ran another extraction today. At this point I just assume it's garbage so I'll be excited when it finally does work. Used a professional kit this time. God damn was that a long procedure. 27 steps, some loop back, and some have up to a 4 hour incubation time. I had to make my own chloroform and find a weird alcohol to make this work. I really hope it just works so I don't need to do that nonsense again. Gonna test it and then try and PCR it in the next couple days if all checks out. Ordered and received the shorter primers that don't have the gibson ends on them. Should increase PCR efficiency a lot. So it looks like things are tentatively stacked in our favor for some success soon. I remain skepitcal though. Might run it on a gel tomorrow just for fun. There's been lots of other side project successes, but will discuss those another time.
-
Test results came back. Got lots of DNA, but when we ran it on a gel it all looks sheared to hell. Still gonna try PCR, but we're starting to suspect that the preservation method of our source spiders isn't good and the DNA degraded. The ethanol prevents them from rotting, but not the nucleases from working. If we're lucky PCR just works and this won't be an issue, but if it is we're gonna need fresher spiders.
-
@chironex Are you gunna smash some fresh spideys or what?